22.09.2024
22.09.2024
In our group, we believe that scientific excellence goes hand in hand with strong management, communication, and leadership abilities. To foster a well-rounded professional development, two of our lab members have recently completed courses designed to enhance these vital skills.
Valentyna Kuznetsova, one of our senior researchers, participated in the esteemed EMBO Laboratory Leadership course, held at the University of South Bohemia. This program, tailored for group leaders, focuses on developing key leadership competencies, including strategic thinking, team management, and conflict resolution. As scientific projects grow in scale and complexity, the ability to lead efficiently and maintain a productive research environment is paramount, making Valentyna’s newly acquired skills important to the success of our team.
In parallel, Alina Kaliuzhna, a new Ph.D. student, attended the Doctoral Summer School at the National Library of Technology (NTK). This two days program equips early-career researchers with critical skills in project management, scientific communication, and interdisciplinary collaboration. Such expertise is essential in navigating the increasingly collaborative and competitive landscape of scientific research, empowering Alina to take on future research challenges with confidence and clarity.
These experiences underscore our lab’s commitment to continuous professional growth, ensuring that our researchers are not only experts in their fields but also effective leaders and communicators.
 22.09.2024
22.09.2024
Ph.D. student Alina Kaliuzhna recently took part in the Summer School at the Extreme Light Infrastructure (ELI) in Szeged, Hungary. ELI ALPS Facility is home to one of the most advanced laser facilities in the world. While there, Alina presented poster on "Dual Photosynthesis: Efficiency of Photosynthetic Pigments in Extreme Conditions," and received valuable feedback from international experts.
In addition to presenting her research, Alina gained practical experience during the school and learned about the latest advances in the field of high-power laser systems. The visit provided her with new skills and insights that she is ready to bring back to her work in the lab.
 22.09.2024
22.09.2024
We are proud to announce that Emrah Özcan has successfully defended his PhD thesis in Biophysics, titled "Tuning Carotenoid Photophysics by Changes in Structure and Local Environment." Emrah’s research focuses on the excited-state dynamics of carotenoids and how they are influenced by external factors like interaction with metal ions, pH changes, external voltage, and chemical modifications.
Earlier this year, in January, Emrah also obtained a Ph.D. in Physical Chemistry from Gebze Technical University in Turkey. This dual expertise has greatly contributed to the depth and innovation of Emrah’s work.
 22.09.2024
22.09.2024
Our long-standing collaboration with the group of Mirek Kloz at ELI Beamlines eventually resulted in two papers, which targeted the vibrational properties of carotenoid excited states in solution and in orange carotenoid protein (OCP). The first paper shows how conformational change of the carotenoid echinenone induced by its binding site in OCP affects the carotenoid vibrations in the excited state, resulting in a specific energy dissipation mechanism in OCP, which could be important for the initial steps of OCP photophysics. The second paper compares the vibrational properties of the lowest excited state of three linear carotenoids, neurosporene, spheroidene, and lycopene. The analysis of vibrations in the S1 state reveals their dependence on the conjugation length of a carotenoid.
 22.09.2024
22.09.2024
Following the previous study, which revealed structure of the OCP-phycobilisome complex and provided a possible explanation of how echinenone in OCP quenches bilins, we have participated in a collaborative project that addresses details of the quenching mechanism. The collaboration was extended to the group of prof. Benedetta Mennucci, who carried out extensive calculations of excited-state properties of echinenone bound to photoactive OCP. The precise calculations were possible due to further improvement of the cryo-EM structure of the OCP-phycobilisome complex. The results, published in Science Advances, showed that specific distribution of charged amino acids in the vicinity of the carotenoid provides a charge distribution that results in a significant enhancement of the transition dipole moment of the S0-S1 transition. This increases coupling between the S1 states of bilins in phycobilisome and echinenone in OCP, opening the energy transfer quenching pathway. Modeling the energy transfer through the whole OCP-phycobilisome complex carried out by our group showed nearly perfect match with the experimental data.
 22.09.2024
22.09.2024
Bis-phenylhydrazone astaxanthin or BPH-astaxanthin in short is a new carotenoid synthesized in our group. For a long time, it is known that conjugated C=O group causes significant changes in excited-state properties of carotenoids. Although keto carotenoids with conjugated C=O group are common in nature and occurs in many photosynthetic organisms, no carotenoids with conjugation groups containing other atom except oxygen exist naturally. Our new synthetic carotenoid replaces two conjugated C=O groups of astaxanthin by C=N groups. Detailed spectroscopic characterization of BPH-astaxanhin has just been published. The new carotenoid is stable in standard organic solvents and has a significantly shorter S1 lifetime than its parent molecule, astaxanthin.
 22.09.2024
22.09.2024
Carotenoids are well known to be strongly hydrophobic molecules insoluble in water, so pH effect has never been studied for carotenoids. However, there are a few exceptions from the “hydrophobicity rule”, such as the hydrophilic carotenoid crocin, known especially as the colorant of saffron. We have made use of the hydrophilic properties of crocin and looked at excited state properties of crocin at different pH values. While acidic pH (<6) destroys the crocin molecule within a couple of hours, crocin is stable at basic pH, allowing spectroscopic experiments up to pH 11. As reported in our paper in PCCP, increasing pH from 7 to 11 induces structural changes reminiscent of those reported upon decreasing of solvent polarity if crocin is dissolved in an organic solvent. Therefore, the results suggest that properties of the conjugated C=O groups of crocin are modified with an increase in pH.
 27.08.2024
27.08.2024
Alina Kaliuzhna, M.Sc. graduated in Biophysics at V.N. Karazin Kharkiv National University. She joined the group in February as a Ph.D. student. She will work within the project focused on investigation of light-harvesting complexes photoprotective functions funded by the Czech Science Foundation.
Alexander Paradzah, Ph.D. joined the group in mid-June. He has previous experience in ultrafast transient absorption spectroscopy gained during his Ph.D. studies at University of Pretoria in South Africa. He will work within the project Photomachines and one of his main tasks will be development of a new experimental setup based on 100 kHz femtosecond laser system.
Shubhankar, M.Sc. joined our group in August. He graduated in Biotechnology at Guru Nanak Dev University in India. He is also a member of the Photomachines project team and will work on preparation, isolation and spectroscopy of the photosynthetic pigments and pigment-protein complexes from microorganisms.
 03.10.2023
03.10.2023
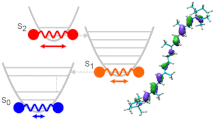
Our research group has just published a new research paper "Time-Resolved Spectroelectrochemical Dynamics of Carotenoid 8'-apo-β-Carotenal" in ChemPlusChem, demonstrating the exceptional ability to fine-tune the amplitude of signals associated with an intramolecular charge transfer (ICT) state of keto-carotenoids through the application of external voltage. Although the spectral position of absorption band remained stable, transient absorption spectra revealed a significant reduction in the magnitude of the ICT-like band during experiments carried out under applied voltage conditions. This phenomenon was accompanied by an extension of the S1/ICT lifetime, changing from 8 ps to 13 ps. Perhaps even more intriguingly, cessation of the applied voltage swiftly led to a return to the no-voltage baseline, demonstrating the reversible nature of our observations. This novel insight into the spectroelectrochemical dynamics of carotenoids opens possible avenues for further exploration of excited state properties of keto-carotenoids.
 31.08.2023
31.08.2023
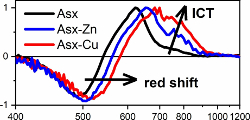
A new study published in the Journal of Photochemistry and Photobiology A: Chemistry reveals the captivating interplay between carotenoid astaxanthin and metal ions. The study's highlights include the formation of stable complexes and their impact on excited-state dynamics. Interaction of astaxanthin with zinc (Zn2+) and copper (Cu2+) ions induces significant alteration of carotenoid excited-state dynamics, resulting in enhancement of the charge transfer character of the lowest excited state, S1/ICT. The study unveils shortened S1/ICT lifetimes of astaxanthin-metal complexes compared to pure astaxanthin due to prolongation of the effective conjugation. The astaxanthin-Cu complex formation also results in observation of the S* signal, which is likely due to a hot ground state generated by rapid depopulation of excited states. This research not only advances our understanding of carotenoid photophysics but also promises possible applications in diverse scientific fields.
 31.08.2023
31.08.2023
A new article "Relaxation dynamics of high-energy excited states of carotenoids studied by UV excitation and pump–repump–probe transient absorption spectroscopy" published in Physical Chemistry Chemical Physics, casts a revealing light on the long-overlooked upper excited states of carotenoids. This research pioneers a comparative investigation into the excited state dynamics of ß-carotene and astaxanthin, uncovering distinct relaxation pathways initiated by direct excitation of the SUV state (observed in absorption spectrum) versus re-pumping of the S1–Sn transition (observed in transient absorption spectrum). The study reveals unique characteristics of the Sn and SUV states and adds further dimension to our understanding of carotenoid photophysics and their roles in biological systems in nature, inviting scientists to explore new realms of discovery across multiple disciplines.
 31.08.2023
31.08.2023
An extensive review paper, coauthored also by a member of our research group, was published in the Chemical Physics Review journal. It unveils the transformative journey of ultrafast laser spectroscopy, from its inception in late 1970s to today's advanced techniques. Titled "Ultrafast laser spectroscopy uncovers mechanisms of light energy conversion in photosynthesis and sustainable energy materials," the paper navigates through decades of ultrafast laser spectroscopy advancements, spotlighting their pivotal role in understanding natural photosynthesis and emerging solar energy materials. Covering topics such as light harvesting, primary electron transfer, and novel solar conversion materials, the review distills the vast landscape of scientific progress to offer a focused insight into critical breakthroughs.
 31.08.2023
31.08.2023
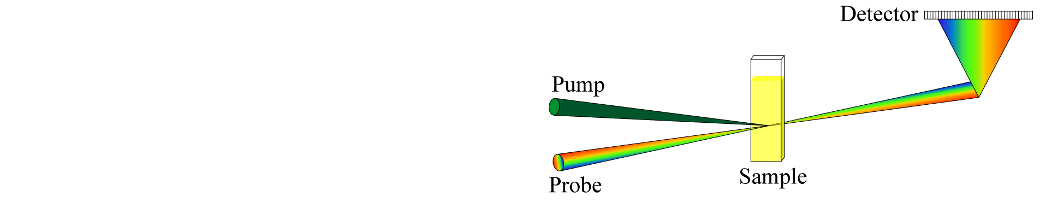
The Laboratory of Optical Spectroscopy is facing a new era of cutting-edge research with a new upgrade to its ultrafast spectroscopy setup. Originally established in 2014 upon our relocation to a new facility, this week marks a pivotal moment as we embark on a journey of modernization and innovation. With a commitment to improve quality of our spectroscopic experiments, our team has disassembled the existing ultrafast spectroscopy setup up to the first mirror, aiming for a new, modern, and compact configuration. This transformative revamp paves the way for enhanced performance and precision, ensuring that our lab remains at the forefront of the field. The upgraded setup presents two distinctive options for white-light probe generation. The first harnesses the power of a fundamental laser beam at 800 nm, while the second leverages near-infrared (NIR) pumped white light generation, covering an expansive spectral window from 400 to 1200 nm. These options open up possibilities for an array of experiments, from pump-probe investigations to multipulse experiments such as pump-dump-probe and pump-repump-probe, as well as two-photon excitation studies. The new and improved ultrafast spectroscopy setup is anticipated to be in full operation mode by the end of September 2023.
 31.08.2023
31.08.2023
The world of carotenoid research converged in a showcase of knowledge exchange and collaboration at the 19th International Symposium on Carotenoids, held in Toyama, Japan from July 9th to 14th. A delegation of researchers from our group, Tomáš Polívka, Vaclav Sebelik, and Valentyna Kuznetsova, have actively participated at the conference, each delivering oral presentations that ignited fruitful discussions amongst their peers. The symposium not only provided a platform for groundbreaking scientific insights but also offered an opportunity for our team to immerse themselves in the vibrant culture of Japan, ensuring an enriching and enjoyable experience throughout their stay.
 25.01.2023
25.01.2023
Energy transfer involving lower excited states of carotenoids and (bacterio)chlorophylls is nowadays well understood topic and we therefore decided to go slightly upwards (on the energy scale). In our study published in Photosynthesis Research, we explored the phenomena following the excitation of the Soret band in LH2 complex from Rbl. acidophilus. Our primary motivation was to find Soret → Car S2 energy transfer but we did not observe any signals attributable to such pathway. On the other hand, our data revealed interesting information about other phenomena taking place in LH2 complex. First, the strong signal in the carotenoid region was modelled as an electrochromic shift resulting from a change of local electric field due to BChl a excitation. Analysis of data further revealed that rhodopin glucoside responds to the excess energy dissipation of BChl a by exhibiting a signal decaying in a few picoseconds exclusively after Soret excitation. Third, we showed that BChl-to-Car triplet-triplet energy transfer took place in 400-600 ps.
 23.12.2022
23.12.2022
We hope that you have a wonderful holiday season, filled with joy, laughter, and time spent with loved ones.
We are looking forward to the new year and all of the exciting discoveries and challenges that it will bring.
Wishing you a Happy New Year,
Polivkalab
 06.09.2022
06.09.2022
Two members of our group, David Bína and Tomáš Polívka participated in a project that resulted in a publication in Nature. The paper, published on 31 August, describes the first complete structure of a cyanobacterial antenna, the phycobilisome. In contrast to expectations, two different conformations of phycobilisomes were identified and characterized. Further, the structure obtained by cryo-electron microscopy allowed us to find and visualize the binding sites of the orange carotenoid protein (OCP), which regulates the energy flow by quenching the antenna under high light conditions. Surprisingly, each phycobilisome can bind four OCPs arranged in two dimers, but the binding is possible only in one of the two phycobilisome conformations. Our task in this project was to model the flow of energy throughout the phycobilisomes with and without OCP. The model clearly showed that OCPs can efficiently quench phycobilisomes by energy transfer from the closest bilins to the carotenoid canthaxanthin bound to OCP.
 04.07.2022
04.07.2022
We are not only presenting our works during scientific conferences, but we also like to participate in events that are aimed at a wider audience and kids. During June the group members became part of the two public events. On the 14th of June, Tomáš Polívka presented a public lecture at Science Café CB titled “What can happen in a trillionth of a second (and how to find out)?” He has shown how we can follow some chemical reactions in real-time and what can happen after light absorption by a molecule. The evening was finalized by open discussion that all participants could enjoy. On the 25 of June, we participated in the MakerDay organized by Objevarium in Ceske Budejovice. Our lab members, Valentyna Kuznetsova and Emrah Özcan, showed some fancy optical tricks to (not only) kids. In collaboration with the Mobile Laboratory, BC AV ČR we have enjoyed the day that exceeded our expectations on public interest in scientific activities.
 20.06.2022
20.06.2022
Our Ph.D. student Ivana Šímová spent three weeks in May at the Technical University of Munich in the group of Prof. J. Hauer as a part of her doctoral internship. Being eager to play with some fancy optics, she spent most of the time together with another Ph.D. student Erika Keil by aligning and optimising a transient absorption setup that uses the achromatic second harmonic generation as a tool to obtain very short and spectrally broad pulses. This complicated task also included a very enjoyable evening session as you can see in the photos below. :)
 20.06.2022
20.06.2022
In our latest paper, we focus mainly on the so-called S* state of carotenoids, routinely detected via the ultrafast spectroscopy for almost 30 years. We present a brief review of this still mysterious state, with the special emphasis on a model based on the vibrational energy relaxation approach (VERA) that aims to explain S* state origin. VERA proved itself to be able to explain the main characteristics of relaxation dynamics after one-photon excitation; nevertheless, the lineshapes after two-photon excitation are still beyond the current model of VERA. In the end of the article, we outline possible future directions in terms of theoretical and experimental methods needed to better describe energy dissipation effects in carotenoids including the first solvation shell.
 21.02.2022
21.02.2022
Following the previous study, we shed the light on light-harvesting complex of G. phototropica again. This member of Gemmatimonadota phylum obtained its photosynthetic genes by a horizontal gene transfer from other bacterial species. Such event allowed G. phototropica to evolve and optimize its own original structure of light-harvesting units, which makes it a very appealing object to study. In a new paper just published in Science Advances, the structure of its light-harvesting complex has been resolved at 2.4 Å by cryo-EM and with the help of ultrafast spectroscopy, the energy-transfer network within the whole photosynthetic unit has been described. It turns out that the elegant double-ring organization of the complex not only provides a very efficient system of energy flow but also demonstrates the endless possibilities of evolution.
 01.12.2021
01.12.2021
Some years back we have identified quenching mechanism in high-light induced proteins (Hlips) from cyanobacteria. The fruitful collaboration with the research group at the Institute of Microbiology of the Czech Academy of Sciences continues. In a new paper just published in Nature Communications, we show that the same quenching mechanism, energy transfer from excited chlorophyll-a to the lowest carotenoid excited state, is also active in two chlorophyll-binding proteins from plants. The Light-harvesting Like (LIL3) protein and Early-Light-Induced Protein (ELIP) bind zeaxanthin, which efficiently quenches excited chlorophyll via energy transfer mechanism. While LIL3 is very efficient quencher, ELIP in its native does not quench, but modification of its N-terminus induces quenching, pointing to the critical role of pigment protein interactions in quenching induction.
 14.10.2021
14.10.2021
Following the successful study of excited states of carotenoids after UV excitation, we have tuned our attention to carotenoids bound to proteins. To simplify the results, we have chosen proteins binding only one carotenoid and no further pigments, the orange carotenoid protein (OCP) and helical carotenoid protein (HCP). In this study, which has just been published in ChemPhotoChem journal, we have used 280 nm excitation to hit both protein (namely Tyr and Trp amino acids) and the carotenoid canthaxanthin, which also mildly absorbs at this wavelength. The results show that UV excitation enhances product formation in OCP in comparison with ‘standard’ excitation by blue-green light. In both OCP and HCP we have identified a canthaxanthin radical cation that is formed with about 5% efficiency exclusively after UV excitation.
 14.10.2021
14.10.2021
After a year without working laser system we are back in full operation. The sad empty space on our optical table is again occupied by (now fully working) MaiTai, thus we spent most of the summer by putting all the pieces of our setup together, upgrading the beamlines and optimizing detection. At the end of July, we eventually run our first experiments after a long break.
 30.3.2021
30.3.2021
Our new paper just published in the Journal of Chemical Physics Letters marks our long and winding road to a functional experimental setup for measurements of transient absorption spectra after two-photon excitation (2PE). After successful test of 2PE transient absorption experiment on carotenoids in solution we reported earlier, this new paper targets a more challenging sample – LHCII antenna from higher plants. By comparing the 2PE transient absorption spectra of LHCII and a mixture of the carotenoid lutein and chlorophyll-a in acetone, we were able to answer the important questions of carotenoid photophysics: Is the carotenoid selectively excited by 2PE or is there also significant contribution to 2PE from chlorophylls? If yes, how large fraction of 2PE photons is actually absorbed by carotenoids? The 2PE transient absorption experiment has proved to be an ideal tool to answer these questions. Our data show that only one third of 2PE photons is absorbed by carotenoids in LHCII, while the dominant contribution to 2PE signal originates from chlorophylls.
 30.3.2021
30.3.2021
There is a number of papers addressing the carotenoid photophysics after excitation into their ‘color-determining’ excited state absorbing in the 400-550 nm spectral range. However, carotenoids also features spectral bands in the 250-330 nm range and whether excitation of these bands somehow changes the excited-state dynamics remains largely unknown. To add this piece of puzzle to the picture of carotenoid photophysics, we have chosen three keto-carotenoids, echinenone, canthaxanthin and rhodoxanthin, and followed their excited-state dynamics in the 400-1200 nm spectral region after excitation of UV absorption bands. The results are summarized in a paper just published in the ChemPhysChem journal. The key conclusion of our experiments is that the enigmatic S* signal is markedly enhanced after UV excitation, underlining its proposed relation to energy dissipation and storage of excess energy in molecular vibrations. We show that the S* signal is not associated with a single state; instead, contributions from both hot S1 state and non-equilibrated ground state forms the S* signal after UV excitation.
 13.1.2021
13.1.2021
 The group members are also involved in teaching and besides teaching university courses and labs,
we often organize lab demonstrations for high school students.
In the section For public, you can watch videos of simple do-it-yourself lab demonstrations using laser pointer as a light source.
The group members are also involved in teaching and besides teaching university courses and labs,
we often organize lab demonstrations for high school students.
In the section For public, you can watch videos of simple do-it-yourself lab demonstrations using laser pointer as a light source.
 3.1.2021
3.1.2021
Although our research primarily focuses on excited-state processes in carotenoids and photosynthetic light-harvesting proteins, time to time we also participate in other projects. Results of such excursion to another research area has been reported recently in Inorganic Chemistry journal. In collaboration with our colleagues from Institute of Inorganic Chemistry of the Czech Academy of Sciences, we studied properties of excited states of boron hydride clusters, compounds promising to serve as an active medium in lasers with emission in the blue spectral region. In a series of alkylated boron hydrides, the data measured in our lab revealed the excited-state absorption band, which in some compounds overlaps with the blue emission band, preventing potential laser action despite high fluorescence quantum yield.
 3.1.2021
3.1.2021
Our group likes to work with ‘exotic’ photosynthetic antenna systems from various microorganisms, which contains unusual carotenoids. One such example is our new paper revealing energy transfer pathways in a membrane-bound antenna from the cryptophyte Rhodomonas salina. This antenna protein has few unusual features. First, it contains both chlorophyll-a and chlorophyll-c (therefore the name CAC antenna), which is combination characteristic for a number of algae, but CAC is antenna is one of a very few containing more chlorophyll-c than chlorophyll-a. Second, CAC binds the carotenoid alloxanthin, the only natural carotenoid containing two triple bonds. Both chlorophyll-c and alloxanthin transfers energy to chlorophyll-a, but efficiencies and time scales differ from ‘standard’ antenna such as LHCII, adding to the diversity of light-harvesting strategies in photosynthetic organisms.
 3.1.2021
3.1.2021
Do you see that empty on our optical table? Anybody who ever worked in a laser lab knows it is unusual to have such a large space without the typical forest of posts and holders of various optical components. Therefore, you may think something strange is going on in our lab. And you are of course correct. This empty space is where our MaiTai laser should be. We were unfortunate to have a serious MaiTai failure during the covid crisis so we had to send it for service to the Spectra Physics labs in Germany. Now the repaired MaiTai is back, but due to travelling restrictions, we are still waiting for a technician to install it. We hope to be ready for new experiments soon. Meanwhile we use this unexpected covid-related break to work on data measured just before the MaiTai failure and even to remove dust from some old data that each of us will certainly find in long-unopened folders in our computers.
 9.10.2020
9.10.2020
We are happy to announce that our colleague Václav Šebelík has successfully completed his PhD work on 24th of September. We were happy to be able to attend his defense in person and strictly followed the recommendations for public events at that time. Though one of the opponents from Germany had to join us virtually due to the travel restrictions. The presentation continued with follow up discussion with the opponents, committee members, and other attendees. After the official part, we had celebrated the end of Vašek’s PhD journey in a slightly new manner. :)
Vašek has started his doctoral studies at the beginning of March 2016 and has quickly became a great fit to our lab. He has been a key person in developing the two-photon and z-scan experimental setups, a perfect partner for any fun or work-related projects in the lab, a “save my time” software developer, as well as our website developer.
 25.8.2020
25.8.2020
Though our research focuses primarily on carotenoids, we have recently diverted our attention to closely related polyenes. Thanks to our collaboration with prof. Ron Christensen from Bowdoin College in Maine, and prof. Tae-Lim Choi from Seoul National University we had opportunity to study two extremely long polyenes having ‘synthetic’ conjugation length N~200. In our new paper published in PCCP, we reported on spectroscopic properties of these polyenes. The experiments were carried at ELI Beamlines laser facility to achieve sub-50 fs time resolution that is not available in our lab. The long polyenes have sub-picosecond lifetime of the S1 state, while the ‘enigmatic’ S* state lasts just slightly longer, 1.8 ps. Comparison of these new data with earlier studies of long polyenes/carotenoids showed that there is essentially no spectroscopic difference between linear conjugated systems having conjugation lengths 50 or 200. Thus, any linear conjugated system with N>50 can be considered as an ‘infinite’ polyene/carotenoid.
 25.8.2020
25.8.2020
HCP’s (helical carotenoid proteins) is a new family of proteins closely related to OCP. Following recent structural characterization of one member of this family, in our recent paper we focused on detailed spectroscopic characterization of two HCP’s, HCP2 and HCP3 binding the carotenoid canthaxanthin. We used our new prism spectrometer to cover whole 400-1200 nm spectral region to capture dynamics of nearly all spectral features in transient absorption spectra. Our data show that both HCP’s exists in two ground state conformations; ‘blue’ conformation excited at 470 nm exhibits an extra band in the S* region, while the ‘red’ conformation excited at 570 nm gives a standard canthaxanthin transient absorption spectrum. For both conformations, the S1 lifetime of canthaxanthin is shorter than in solution, reflecting the twist of one of the terminal rings revealed by the X-ray structure. Despite detailed structural and spectroscopic characterization, the function of HCP’s in cyanobacteria remains unknown.
 25.8.2020
25.8.2020
In a joint effort with the group of Alexander Ruban from the Queen Mary University in London, we have taken a slightly different approach to explore mechanism of non-photochemical quenching in LHCII. It is well-known fact that quenching can be induced by aggregation of LHCII complexes and, when working at low detergent conditions, it is nearly impossible to prevent this aggregation. In experiments described in a new paper in iScience, we have immobilized LHCII trimers in polyacrylamide gel in both quenched and unquenched conformation to prevent aggregation. Under these conditions, the aggregation-induced quenching is minimal and the observed quenching must be related to specific conformation of individual non-interacting LHCII trimers. Transient absorption experiments on LHCII in gel revealed a new carotenoid spectral band whose amplitude is related to quenching.
 4.6.2020
4.6.2020
Following our recent discovery that non-conjugated acyloxy groups can essentially switch off the intramolecular charge transfer (ICT) state of fucoxanthin if it is at the opposite side of the molecule as the conjugated keto group, we have been searching for a keto-carotenoid, which would have the acyloxy group close to the conjugated keto group. Indeed, there exists a pair of keto-carotenoids, siphonaxanthin and siphonein, that have either no acyloxy group (siphonaxanthin) or an acyloxy group at the same side of the molecule as the keto-group (siphonein). In our new paper recently published in Photosynthesis Research we show that the “acyloxy switch” really works. Depending on the position of the acyloxy group in respect to the conjugated keto group, it can either switch off (acyloxy opposite to keto) or switch on (acyloxy at the same side as keto) the ICT state.
 1.4.2020
1.4.2020
The coronavirus infection touches the work of all of us. Yet, thanks to modern technologies and young group members who know how to use them effectively we have organized a virtual group meeting. Obviously, the spirit of the group is not much affected by the current situation. We will continue this way of communicating the results until we can go back to our offices and labs and have standard group meetings.
 1.4.2020
1.4.2020
A new paper from the group has just appeared online in Photochemical and Photobiological Sciences. Thanks to our colleagues from the University of Cagliari and from the Warsaw University of Life Sciences we got a carotenoid-binding protein that has not yet been studied by ultrafast spectroscopy. It comes from the bacterium Deinococcus radiodurans that is well-known for its ability to sustain high doses of UV radiation essentially without damage. The bacterial cell wall of the bacterium is coated by the S-layer, which contains highly ordered two-dimensional arrays of proteins, which form hexameric complexes binding the carotenoid deinoxanthin (S-layer Deinoxanthin Binding Complex – SDBC). The paper provides the first detailed spectroscopic characterization of this carotenoid, both in solution and bound to SDBC.
 2.3.2020
2.3.2020
Our fresh Ph.D. student, Ivča Šímová, has just returned from the workshop “Optimization of light energy conversion in plants and microalgae”, organized under SE2B in Porto, Portugal. She has presented the first data from her Ph.D. project focused on energy transfer processes in unusual purple bacterial antenna. The recently described antenna system from Gemmatimonas phototrophica featuring antenna having two concentric rings of BChl-a, and so far unknown carotenoid is just such unusual antenna.
 2.3.2020
2.3.2020
After publishing two papers on pump-dump-probe spectroscopy in 2018, we took a short break as the two main persons behind the pump-dump-probe setup (Robert and Valja) left the group. Now, when Valja is back after nearly two-year postdoc stay in the group of Janne Ihalainen in Jyvaskyla, we are reviving the PDP setup again with a hope to reveal new features of excited state dynamics of those fantastic orange molecules – carotenoids. In the new detection setup, the PDP experiment is coupled with the prism spectrometer so we can see the effect of dumping/re-pumping in much broader spectral window than two years ago.
 24.2.2020
24.2.2020
The February issue of BBA-Bioenergetics published our latest contribution to research of OCP and related proteins. This time we join efforts with our colleagues from other labs to combine spectroscopy and structural analysis of two OCP forms, OCP1 and OCP2. From the spectroscopic point of view, the paper shows our first data measured with a prism spectrometer in the detection system allowing to measure transient absorption spectra in the 400-1200 nm spectral region. The data presented in the paper were collected using the setup of our friends at ELI Beamlines, but now we have our own detection with prism spectrometer to run the experiments – our own data will appear soon elsewhere! Of course, we need to give up some spectral resolution in such experimental arrangement, but to see all spectral and dynamical features in the whole 400-1200 nm spectrum is simply fantastic. In the figure bellow you can see an example of the super broadband white-light continuum from our lab generated in a sapphire plate as it is seen at the CCD detector after the prism spectrometer.
 18.12.2019
18.12.2019
The laboratory of Optical Spectroscopy has been founded in 2005 at the Institute of Physical Biology based in Nové Hrady, University of South Bohemia in České Budějovice. December 2013 we have relocated the laboratory to the newly built university facility at the Faculty of Science, University of South Bohemia in České Budějovice. Since then, we had shared sextillions of femtoseconds of happy and exciting moments, and had lightened up many of the research ideas. Here is the short summary of the recent years in photos of social events...